
The atomic mass is the relative mass of an element, while the atomic number is a unique identifier for each element. ConclusionĪtomic mass and atomic number are two important components of an atom that help to identify it. However, its actual atomic number is 16 – remember, this refers to the total number of protons in the nucleus, not just those in the outermost shell! The extra protons are located in 2 inner shells thus, oxygen has 2 more electrons than protons. You might think that since oxygen has 8 protons, it would have an anatomic number of 8. The periodic table lists elements in order of increasing atomic number. It determines what element an atom is.įor example, all atoms with an atomic number of 1 are hydrogen atoms those with an atomic number of 2 are helium atoms, and so on. The atomic number is simply the number of protons in an element’s nucleus. The masses of other atoms are then measured relative to this Carbon-12 scale.įor example, the oxygen atom has an atomic mass of 16 Da, which means it is slightly heavier than carbon. This means that the atomic mass of a carbon-12 atom is 12 Da. Atomic mass is measured in daltons (Da), which is defined as 1/12th of the mass of a carbon-12 atom. There is a difference between atomic mass and atomic number, even though they both pertain to atoms.Ītomic mass is the total mass of an atom, while atomic number is the number of protons in an atom.īoth are important when studying chemistry and other sciences that deal with atoms. Difference between Atomic mass and Atomic number The relative atomic mass of an element can be determined by finding the average mass of all the isotopes of that element. The standard for comparison is carbon-12, which has a mass of 12 atomic units. The relative atomic mass of an element is the mass of one atom of that element, relative to the mass of atoms of other elements. To find an element’s Atomic mass, simply add together the number of protons and neutrons in the nucleus of an atom of that element. The Atomic mass is the sum of the protons and neutrons in an atom. To calculate Atomic mass, you need to know the element’s Atomic number and the element’s mass number. The atomic number is the number of protons in an atom. The atomic mass is the sum of the masses of the protons and neutrons in an atom. Define Atomic massĪtomic mass is the mass of an atom, typically given in units of atomic mass units (amu). 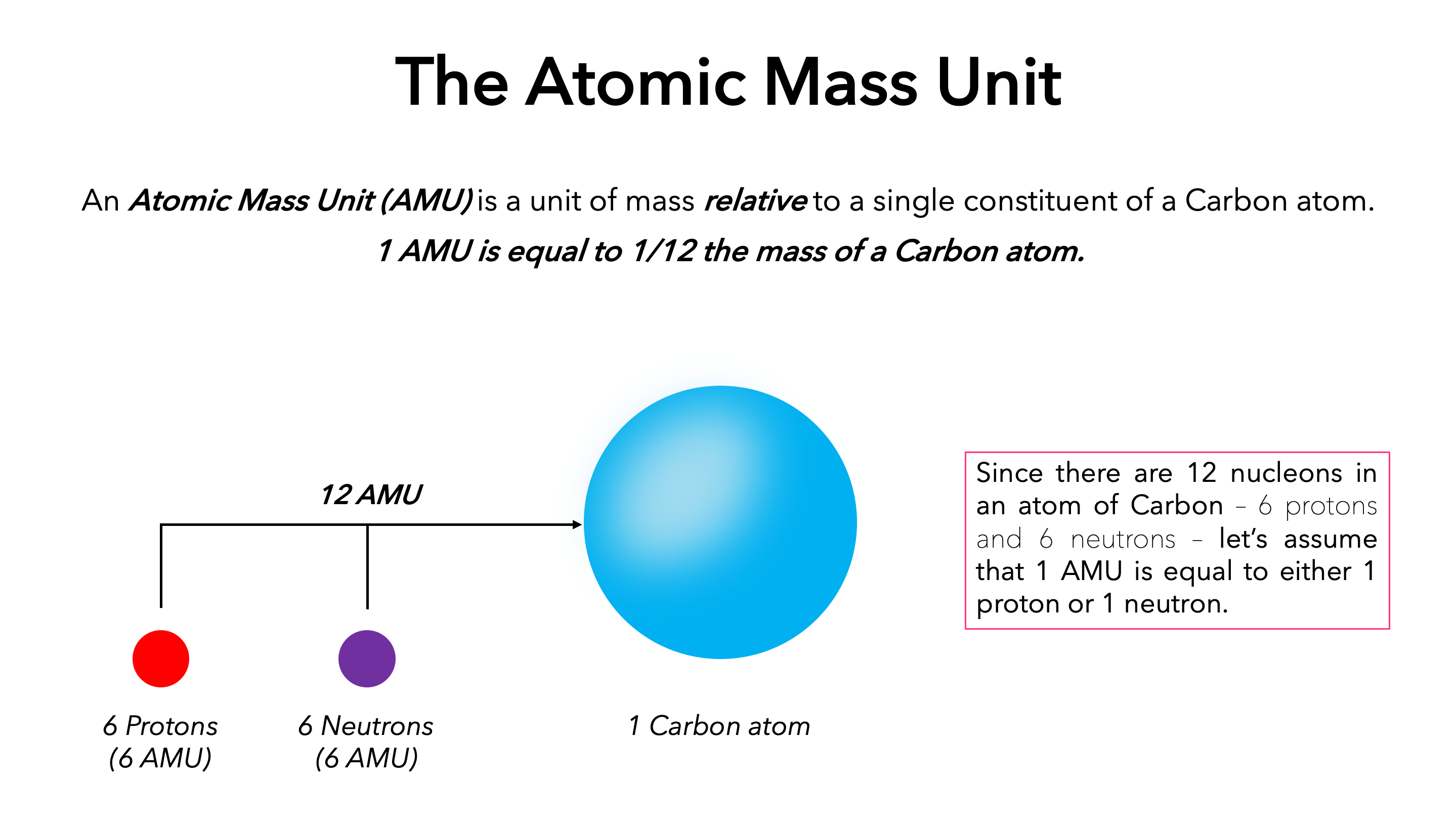 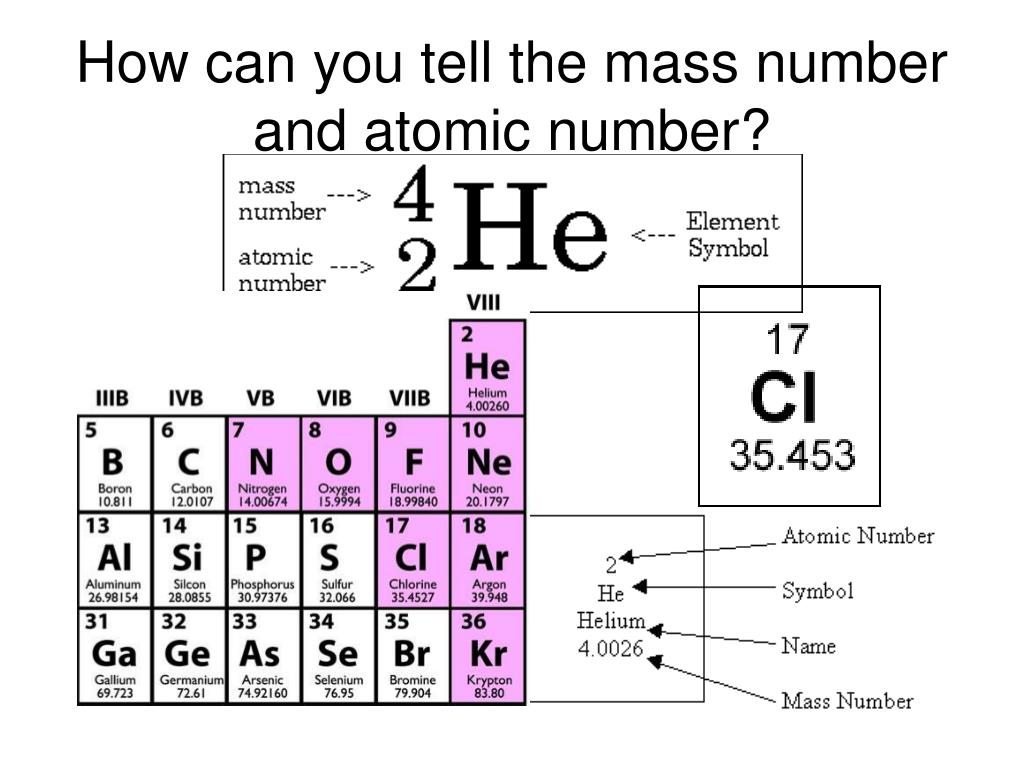
To get a better understanding of what these numbers mean, let’s dive into what sets them apart as well as how they can be used to identify elements. These two values provide essential information about the atom, but their differences are sometimes overlooked. Now imagine that you could somehow keep dividing the speck of carbon into smaller and smaller pieces.When studying elements, it’s important to understand the differences between atomic mass and atomic number. Each speck of carbon would still have all of the physical and chemical properties of carbon. Imagine taking a small piece of carbon and grinding it until it is a fine dust. The graphite in your pencil is composed of the element carbon, a nonmetal. Carbon-13 behaves chemically in exactly the same way as the more plentiful carbon-12. Chemical behavior is governed by the number of electrons and the number of protons. While the presence of isotopes affects the mass of an atom, it does not affect its chemical reactivity. The nucleus of a given carbon atom will be one of the three possible isotopes discussed above. Tin has ten stable isotopes, which is the most of any known element. Carbon has only three natural isotopes, but some heavier elements have many more. Naturally occurring samples of most elements are mixtures of isotopes.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
